Influenza Vaccine Coverage and Efficacy among King Salman Armed Forces Hospital 2017-2018
Salem K Al Dahi, Rofayda M Ahmed, Shatha T Alhakami, Eman Gashgary, Sara A Alaeed, Maryam H Salami and Sahar H Salami
DOI10.21767/2572-5483.100042
Department of Preventive Medicine, King Salman Armed Forces Hospital, Tabuk City, Saudi Arabia
- *Corresponding Author:
- Rofayda M Ahmed
Department of Preventive Medicine
King Salman Armed Forces Hospital
Tabuk City, Saudi Arabia
Tel: +0545450874
E-mail: rafimedcine@gmail.com
Received date: July 08, 2018; Accepted date: October 05, 2018; Published date: October 12, 2018
Citation: Dahi SKA, Ahmed RM, Alhakami ST, Gashgary E, Alaeed SA, et al. (2018) Influenza Vaccine Coverage and Efficacy among King Salman Armed Forces Hospital 2017-2018. J Prev Med Vol.3 No.3:21.
Copyright: © 2018 Dahi SKA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Despite recommendations for annual influenza vaccination of health care workers (HCWs) still vaccination coverage is low, studies suggested that misconceptions about safety or efficacy discourage HCWs from up taking the vaccine.
Methods: This is a case control study to measure coverage and efficacy of seasonal influenza vaccination among HCW at king Salman Armed Forces Hospital-Saudi Arabia 2017-2018. Case group were vaccinated (324) and control group were not (324).
Results: Total number of employee 4118, Influenza Vaccination coverage among employee, non-medical and medical are 45.11%, 34.76% and 61.73% respectively. The results showed higher acceptance rate among female than male, higher among non-Arabic nationality next, non-Saudi Arabic then Saudi nationality but did not show relationship with age or smoking. Comparison of the two groups for vaccine efficacy failed to report any association between vaccination status and incidence of influenza like illness, pneumonia, sever acute respiratory infection, hospital admission, seeking medical care or even decreasing duration of sick leaves. Willingness to uptake the coming vaccine found to be associated with up taking the current one and no association with being infected during this season.
Conclusion: This study revealed vaccination acceptance rate increases with being male, non-medical staff Non- Arabic and Non-Saudi Arabic nationality rather than Saudi one, no relationship with age group and smoking. However, failed to show association between up taking the vaccine and lowering incidence of influenza like illness, pneumonia, sever acute respiratory infection, hospital admission, seeking medical care or even decreasing duration of sick leaves. We suggested that vaccine-targeted viruses are not compatible with the circulating viruses at Tabuk area, so studies are needed to identify these viruses.
Keywords
Coverage; Efficacy; Seasonal; Influenza; Infection; Vaccination; HCW
Introduction
A well established fact in modern medicine; immunization has had the most profound impact in saving lives and in preventing morbidity and disability. Vaccination of healthcare workers (HCW) against influenza is a mode of infection control in healthcare settings.
In spite of the recommendation of the CDC Healthcare Infection Control Advisory Committee and the Advisory Committee on Immunization Practices regarding provision of influenza vaccination to HCWs at the work site, free of charge, as an infection control measure, still the rate of vaccination uptake by HCWs is low. According to a recent analysis of data from the 2000 National Health Interview Survey in which 1651 HCWs were included within 12 months of survey, the vaccination rate in the United States was reported to be only 38%. Furthermore, a recent studies also concluded that despite recommendations, less than 25% of HCWs in Europe and the United Kingdom are vaccinated against influenza.
The benefits of vaccination and protection of staff against influenza infection include prevention of transmissions to patients and the reduction of economic loss due to staff absenteeism. However, doubts about the efficacy and necessity of influenza vaccination are common. To increase the vaccination rate, HCWs need to be convinced about the efficacy and safety of the vaccine. The current study measuring the coverage and efficacy of influenza vaccination among healthcare worker at King Salman Armed Forces Hospital.
Project Objectives
To measure coverage and efficacy of seasonal influenza vaccination among healthcare worker at King Salman Armed forces hospital in Tabuk City 2017-2018, by comparing vaccinated and non-vaccinated groups.
Literature survey
Seasonal influenza is a major public health challenge. Mortality rate is 3–5 million cases yearly [1]. All age groups are affected especially elderly, annual attack rates average 10 to 20 percent [2]. Influenza is sometimes associated with malaise persisting for several weeks and often results in restriction of activity [3]. Infected persons shed virus for 24 hours prior to the development of symptoms, up to a week after recovery [4]. Influenza accounts for millions of days lost from work each year [5].
Nosocomial influenza is regarded as an emerging issue, especially among immunocompromised patients [6]. Hospitalacquired influenza (HAI) showed significant morbidity and mortality in hospitalized patients [7]. Vaccination can also reduce sick leave and provide economic benefits for healthcare institutions [8].
HCW vaccination has been associated with reductions in nosocomial infections [9] Prevention and control of nosocomial influenza entail multiple measures; vaccination of healthcare workers (HCW) is advocated by the Centers for Disease Control and Prevention to obviate influenza transmission in healthcare settings [10].
There are strong scientific evidences regarding influenza vaccination efficacy and policies that promote immunization, but still coverage rates remain low according to reports from the United States; 66.9%, [11] Brazil [12] (43.2%), Australia [13] (16.3-58.7%), European countries like Spain [14] (maximum 26.3%). One study conducted at WHO showed 57.7% of UK healthcare workers would accept the pre-pandemic H5N1 vaccine, [15] so additional data are needed to establish the benefits of vaccination and promote it among HCW and more aggressive interventions, such as implementing mandatory influenza vaccination policies, are needed to achieve higher vaccination rates [16].
The current recommendations of the Advisory Committee on Immunization Practices focusing on persons at increased risk for complications [17].
Modelling studies have shown that vaccination reduces infection, hospitalization, mortality and morbidity [18]. Since the supply of vaccines will be limited, prioritization in the administration is essential [19]. In nearly all countries, healthcare workers are listed as the priority group for mass vaccination [20]. Also studies have shown that HCW vaccination rate is linked linearly to the influenza attack rate in nursing homes and [21] acute-care hospitals [22]. Nosocomial infections increase organizations burden to ensure patient safety and limit the cost of hospitalization [23].
According to the literature, major causes of vaccination rejection in HCW are “concerned about side effects, believed vaccine is not safe” [24]. Others have doubts about its efficacy [25]. There is lack of knowledge among HCW about the vaccine [26]. Therefore, they do not advise their patients to vaccinate [27]. To encourage HCW vaccination there are programs that include free vaccination, education to address misconceptions, the need and benefits, management support and the use of declination forms have been shown to increase coverage [28]. Because with voluntary uptake the rate remains low [29], Debate continues and some authors’ recommended that influenza vaccine should be mandatory in every HCW with patient contact [30].
Vaccination used in this study was Vaxigrip, which is split virion, inactivated, it is propagated in fertilized hen egg and hemagglutinin, and it complies with the WHO recommendations (Northern Hemisphere) and EU decision for the 2017/2018 season [31].
Study design
This is a case control study to measure vaccine coverage and to compare case group subjects who received seasonal influenza vaccine and those in the control group who did receive neither vaccine nor placebo.
Methods
The study was conducted at King Salman Armed Forced Hospital located in North Western Area of Saudi Arabia 2017-2018, the eligibility criteria is to be employed full-time, and had no medical conditions, such as chronic cardiopulmonary disease, diabetes mellitus, or other serious medical conditions, that would place them at high risk for complications of influenza. The criteria for exclusion were; pregnancy and history of immediate hypersensitivity reactions to eggs (because the vaccine may contain small amounts of residual egg protein, as thimerosal is the preservative in the vaccine). Informed consent was obtained from all participants. The study was approved by the research ethical committee of hospital.
The study population included all hospital employees who were vaccinated during this season at the beginning of September 2017; by calculation, resulted sample size was 319.
There are efforts to foster vaccination like face-to-face communication and an announcement posted on the hospital intranet for all personnel. In addition, a more active vaccination strategy was designed as campaign, consisting of information and training for hospital staff in the form of speeches, posters. The refusal forms were not used in the influenza vaccination program, because such a practice is not common in Saudi Arabia for this type of vaccination.
Data collection methods, instruments used, measurements
Variables measured in this study were: Vaccination coverage, Gender, Age, Department, Smoker or not, History of pneumonia, Developing Influenza Like Illness, Developing Sever Acute Respiratory Infection, Absence from work, Duration of illness, Seeking medical care, Hospital addition and willingness to up take the coming influenza vaccine.
Statistical analysis
Descriptive statistics was used to describe the data. For categorical variables, frequencies for case and control groups were compared by chi square test as all variables used were categorical and all statistical analysis was performed with SPSS 21 for Windows software. Data entry, analysis and result interpretation was done by researchers.
Ethical considerations
The study was approved by the ethical committee of the hospital; Verbal informed consent was obtained from participants involved in the study prior to questionnaire administration. Allocation of participants in case or control group depended on his vaccination status which is personal choice in spite of all fostering regulations.
Results
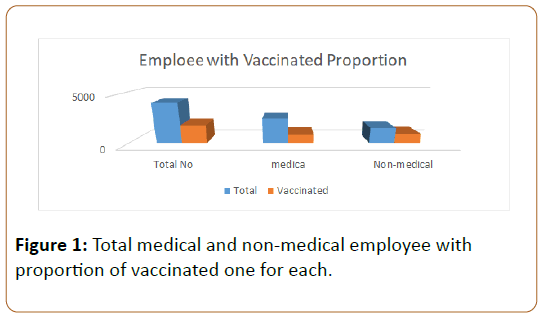
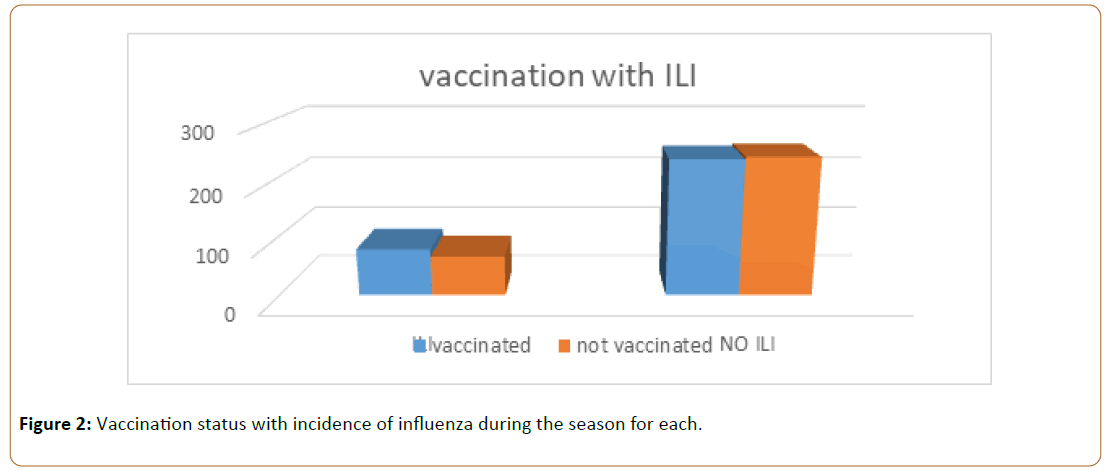
As shown in Figure 1 and Figure 2 the total number of employee at King Salman Armed Forced Hospital located in North Western Area of Saudi Arabia 2017-2018, is 4118 of them vaccinated are 1858 representing (45.11%), medical staff are 2537 of them 882 representing (34.76%) are vaccinated, Non- Medical staff are 1581 of them 976 representing (61.73%) are vaccinated, the difference between them is 28.03% and 95% confidence interval (25.03-31.03) as it doesn’t contain zero so it is significant. By calculation sample size was 319 we took 324 vaccinated as a case group, they were selected by simple randomization and compared by 324 who were not vaccinated as a control, so both case and control groups were exposed to similar virological strains across time.
The demographic characteristics of these subjects, reported in Table 1, their ages are distributed into three categories. Vaccine coverage was 45% for all of the staff, 34.7% for medical and 61.7% for non-medical staff. Table 1 reports the demographic characteristics of cases and controls groups.
| Age categories | vaccinated | Not vaccinated | χ2-value* (P-value) |
|---|---|---|---|
| Less than 30 | 118 (18.2%) | 118 (18.2%) | 0.91 |
| 30-50 | 180(27.7%) | 177 (27.3%) | 0.91 |
| More than 50 | 26 (4%) | 29 (4.45) | |
| Gender | |||
| male | 158(24.38%) | 193(29.78%) | 0.006 |
| female | 166(25.61%) | 131(20.21%) | 0.007 |
| Nationality | |||
| Saudi | 119 (18.36%) | 217 (33.48%) | 0 |
| Arabic Non-Saudi | 29 (4.47) | 21 (3.24%) | 0 |
| Non-Arabic | 176 (25.77%) | 86 (31.28%) | |
| Departments according to the degree of risk exposure | |||
| High Risk | 39 (6%) | 31 (4.78%) | 0.242 |
| Risk | 47 (7.25%) | 65 (10.03%) | 0.241 |
| Low risk | 68 (10.49%) | 69 (10.64%) | |
| 170(26.23%) | 159 (24.53%) | ||
| Smoker or not | |||
| 65 (10.03%) | 80 (12.34%) | 0.157 | |
| Not Smoker | 259 (39.96%) | 244 (37.65) | 0.187 |
Table 1: Demographic and clinical variables.*Chi-square test
Discussion
The is a prospective study design to measure seasonal influenza vaccine coverage and efficacy among healthcare worker at King Salman Armed Forced Hospital located in North Western Area of Saudi Arabia 2017-2018, in similar flu season, so both case and control groups were exposed to similar vaccine and virological strains across time. There is a previous study identifying the circulating strains in Jeddah–Saudi Arabia: A/ pdmH1N1 virus, A/H3N2 virus, and B Yamagata-like virus [32]. But there is no similar previous study in Tabuk area. The vaccine used was Vaxigrip which is intended to protect against the three strains of the vaccine.
Vaccine coverage was 45% for all of the staff, 34.7% for medical and 61.7% for non-medical staff; this result revealed that coverage is higher in non-medical staff than the medical and the difference is statistically significant, Medical staff is responsible for encouraging their patients to get the vaccine, a previous study in five European countries during season 2006/07 revealed that: The major factor for vaccination (61%) was a recommendation by the family doctor or nurse [33]. The lower coverage rate with the medical staff needs to be investigated by further studies.
Table 1 reported the demographic characteristics of cases and controls groups: Their ages are distributed into three categories and there is no significant association between participants age category and vaccination status. Regarding gender distribution between the two groups females are more representative in the case (vaccinated group) and the difference is statistically significant. In addition, the results revealed significant relationship between nationality and up taking influenza vaccine with higher probability of non-Arabic nationality next, non-Saudi Arabic then Saudi nationality.
Departments were ranked according to exposure to infected patients into high-risk departments, moderate and low risk then the non-medical staff; but failed to find any relationship with being vaccinated, these results are not consistent with previous study done in Riyadh in multi-nationality health-care workers in Saudi Arabia 2009 found that Important factors associated with increasing influenza vaccine acceptance include being a male, Non-Saudi Arabic nationality, In addition to working in departments at high risk of exposure to influenza viruses [34].
Even for smoker and non-smoker no detected relationship, which is consistence with the results of a study aimed to determine reasons for not complying with vaccination among smokers and non-smokers (n=4000 Dutch participants) revealing that there are no substantial differences in complying with influenza vaccination between smokers and non-smokers [35]. Table 2 showed Chi square testing association between vaccination status and incidence of influenza like illness, pneumonia, sever acute respiratory infection; which failed to find significant association. Considering that both case and control groups were exposed to similar virological strains during the season, Many studies confirmed that if Influenza is circulating in a community, the presence of cough and high fever in a patient is likely to be associated with influenza [36]. Table 3 and Table 4 demonstrating the relationship between vaccination status and variables that indicating illness severity: It did not show statistically significant differences between the two groups for duration of the illness, absence due to the illness, seeking medical care, hospital admission or taking Medication.
| Incidence of Pneumonia | vaccinated | Not vaccinated | χ2-value* (P-value) |
|---|---|---|---|
| Yes | 27 (12.80%) | 21 (3.24%) | 0.368 |
| No | 297 (45.83%) | 303 (46.75%) | 0.453 |
| Influenza Like Illness | |||
| Yes | 83 (14.66%) | 70 (10.80%) | 0.229 |
| No | 241 (37.19%) | 254 (37.80%) | 0.227 |
| Sever Acute Respiratory Infection | |||
| Yes | 12 (1085%) | 21(3.24%) | 0.108 |
| No | 312 (48.14%) | 303 (46.75%) | 0.106 |
Table 2: Incidence of illness during the season by vaccinated and not vaccinated group.
| Duration of the illness | vaccinated | Not vaccinated | χ2-value* (P-value) |
|---|---|---|---|
| Non | 288 (44.44%) | 244 (37.65%) | 0.293 |
| Less than 2 days | 86 (13.27%) | 69 (10.64%) | 0.293 |
| 2 days or more | 10 (1.54%) | 11 (1.69%) | |
| Absence due to the illness | |||
| Yes | 12 (1.85%) | 21 (3.24%) | 0.108 |
| No | 312 (48.14%) | 303 (46.75%) | 0.106 |
| Seeking medical care | |||
| Yes | 12(1.85%) | 21(3.24%) | 0.108 |
| No | 312(48.14%) | 303(46.75%) | 0.106 |
| Hospital Admission | |||
| Yes | 9 (1.38%) | 7 (1.08%) | 0.033 |
| No | 315 (48.61%) | 317 (48.91%) | 0.027 |
| Taking Medication | |||
| Yes | 98 (15.12%) | 79 (12.19%) | 0.094 |
| No | 226 (4.01%) | 245 (37.80%) | 0.113 |
Table 3: Indicators of illness severity, by vaccinated and not vaccinated group.
| Will take the vaccine and vaccination status | vaccinated | Not vaccinated | χ2-value* (P-value) |
|---|---|---|---|
| Yes | 202 (31.17%) | 105(16.20%) | 0 |
| No | 122 (18.82%) | 219 (33.37%) | 0 |
| Developing illness and willingness | |||
| Yes | 84 (12.96%) | 82 (12.65%) | 0.334 |
| No | 233 (35.95%) | 259 (39.96%) | 0.382 |
Table 4: Willing to take the coming year vaccine.
These results are not consistent with many studies evaluated the efficacy of this vaccine on the rate of hospitalization for influenza and for complications of influenza, including pneumonia, all acute and chronic respiratory conditions, its effect on the costs of hospitalization and medication [37].
Regarding willingness to uptake the vaccine for the coming season the results showed significant relationship with up taking the current vaccination, but failed to find association with getting infected during this season, similar study found that the willingness to accept influenza vaccination among hospital based healthcare workers in Hong Kong was low and the strongest associations with the intention to accept vaccination were a history of seasonal influenza vaccination.
Conclusion
The study found influenza vaccine acceptance is higher among non-medical staff, there are important factors associated with increasing probability of vaccination include being a male, Non- Arabic and Non-Saudi Arabic nationality, no relationship with age group and smoking, regarding vaccine efficacy the result failed to show association between up taking the vaccine and low incidence of influenza like illness, pneumonia, sever acute respiratory infection, hospital admission, seeking medical care or even decreasing duration of sick leaves. We suggested that the vaccine-targeted viruses are not compatible with the circulating viruses of Tabuk area, so further studies are needed to identify these viruses.
References
- https://www.who.int/mediacentre/factsheets/fs211/en/index.html
- LaForce FM, Nichol KL, Cox NJ (1994) Influenza: Virology, epidemiology, disease and prevention. Am J Prev Med 10: 31-44.
- Sullivan KM, Monto AS, Longini Jr IM (1993) Estimates of the US health impact of influenza. Am J Public Health 83: 1712-1716.
- Carrat F, Vergu E, Ferguson NM, Lemaitre M, Cauchemez S, et al. (2008) Time lines of infection and disease in human influenza: A review of volunteer challenge studies. Am J Epidemiol 167: 775-785.
- Riddiough MA, Sisk JE, Bell JC (1983) Influenza vaccination: Cost-effectiveness and public policy. JAMA 249: 3189-3195.
- Salgado CD, Giannetta ET, Hayden FG, Farr BM (2004) Preventing nosocomial influenza by improving the vaccine acceptance rate of clinicians. Infect Control Hosp Epidemiol 25: 923-928.
- Cella MT, Corona G, Tuccillo E, Franco G (2005) Assessment of efficacy and economic impact of an influenza vaccination campaign in the personnel of a health care setting. Med Lav 96: 483-489.
- Salgado CD, Farr BM, Hall KK, Hayden FG (2002) Influenza in the acute hospital setting. Lancet Infect Dis: 145-155.
- Fiore AE, Shay DK, Broder K, Iskander JK, Uyeki TM, et al. (2009) Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009. MMWR Recomm Rep 58: 1-52.
- Influenza vaccination coverage among healthcare personnel 2011-2012 influenza seasons, United States. MMWR 61: 753-757.
- De Souza CR, Zambonatto JAM, Bastelo Branco FC (2010) Predictors of adherence to influenza vaccination for healthcare workers from a teaching hospital: A study in the prepandemic era. Rev Soc Bras Med Trop 43: 611-614.
- Seale H, MacIntyre CR (2011) Seasonal influenza vaccination in Australian hospital health care workers: A review. Med J Aust 195: 336-338.
- Picazo JJ, Alonso LM, Arístegui J, Bayas JM, Sanz J, et al. (2012) Consensus on vaccination against influenza in health personnel. Spanish Journal of Chemotherapy 25.
- Van den DC, Bonten MJ, Hak E, Heijne JC, Wallinga J, et al. (2008) The effects of influenza vaccination of health care workers in nursing homes: Insights from a mathematical model. PLoS Med 5: e200.
- Ajenjo MC, Woeltje KF, Babcock HM, Gemeinhart N, Jones M, et al. (2010) Influenza vaccination among healthcare workers: Ten year experience of a large healthcare organization. Infect Control Hosp Epidemiol 31: 233-240.
- Nichol KL, Margolis KL, Wuorenma J, Von Sternberg T (1994) The efficacy and cost effectiveness of vaccination against influenza among elderly persons living in the community. N Engl J Med 331: 778-784.
- Nuno M, Chowell G, Gumel AB (2007) Assessing the role of basic control measures, antivirals and vaccine in curtailing pandemic influenza: Scenarios for the US, UK and the Netherlands. J R Soc Interface 4: 505-521.
- European centre for disease prevention and control (2007) technical report: Expert advisory groups on human H5N1 vaccines.
- Ministry of health NZ.H5N1 pre- pandemic vaccine consultationdocument 2007 10/Traynor K: United State issue draft guidance for pandemic flu vaccination. Am J Health Syst Pharm 64: 2412-2414.
- Van den DC, Bonten MJ, Hak E, Wallinga J (2009) Modeling the effects of influenza vaccination of health care workers in hospital departments. Vaccine 27: 6261-6267.
- Maltezou HC (2008) Nosocomial influenza: New concepts and practice Curr Opin Infect Dis 21: 337-343.
- Ducel G, Fabry J, Nicolle L (2002) World Health Organization. Prevention of hospital acquired infections: A practical guide.
- Barriere J, Vanjak D, Kriegel I, Otto J, Peyrade F, et al. (2009) Acceptance of the 2009 A (H1N1) influenza vaccine among hospital workers in two French cancer centers. Vaccine 28: 7030-7034.
- Savas E, Tanriverdi D (2010) Knowledge, attitudes and anxiety towards influenza A/H1N1 vaccination of healthcare workers in Turkey. BMC Infect Dis 10: 281.
- Torun SD, Torun F, Catak B (2010) Healthcare workers as parents: Attitudes toward vaccinating their children against pandemic influenza A/H1N1. BMC public health 10: 596.
- Abramson ZH, Levi O (2008) Influenza vaccination among primary healthcare workers. Vaccine 26: 2482-2489.
- Tapiainen T, Bär G, Schaad UB, Heininger U (2005) Influenza vaccination among healthcare workers in a university children's hospital. Infect Control Hosp Epidemiol 26: 855-858.
- Van Delden JJ, Ashcroft R, Dawson A, Marckmann G, Upshur R, et al. (2008) The ethics of mandatory vaccination against influenza for health care workers. Vaccine 26: 5562-5566.
- Poland GA (2010) Mandating influenza vaccination for health care workers: Putting patients and professional ethics over personal preference.
- Blank PR, Schwenkglenks M, Szucs TD (2008) Influenza vaccination coverage rates in five European countries during season 2006/07 and trends over six consecutive seasons. BMC public health 8: 272.
- Tolah AM, Azhar EI, Hashem AM (2016) Susceptibility of influenza viruses circulating in Western Saudi Arabia to neuraminidase inhibitors. Saudi Med J 37: 461.
- Puig BJ, Guglieri LB, Tortajada GM, López LFX, Carballido FM, et al. (2017) for the Study VH. low influenza vaccine effectiveness and the effect of previous vaccination in preventing admission with A (H1N1) pdm09 or B/Victoria-Lineage in patients 60 years old or older during the 2015/2016 influenza season. Vaccine 35: 7331-7338.
- Al Tawfiq JA, Antony A, Abed MS (2009) Attitudes towards influenza vaccination of multi nationality health care workers in Saudi Arabia. Vaccine 27: 5538-5541.
- Looijmans van DAI, Van Den HPM, Verheij TJ, van Delden JJ, Van Essen GA, et al. (2007) No intention to comply with influenza and pneumococcal vaccination: behavioural determinants among smokers and non-smokers. Preventive medicine 45: 380-385.
- Boivin G, Hardy I, Tellier G, Maziade J (2000) Predicting influenza infections during epidemics with use of a clinical case definition. Clin Infect Dis 31: 1166-1169.
- Nichol KL, Margolis KL, Wuorenma J, Von Sternberg T (1994) The efficacy and cost effectiveness of vaccination against influenza among elderly persons living in the community. N Engl J Med 331: 778-784.
- Chor JS, Ngai KL, Goggins WB, Wong MC, Wong SY, et al. (2009) Willingness of Hong Kong healthcare workers to accept pre-pandemic influenza vaccination at different WHO alert levels: Two questionnaire surveys. BMJ 339: b3391.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences