The Accuracy of a Volume Plethysmography System as Assessed by Contrast Angiography
Andrew Gomes
DOI10.21767/2572-5483.100036
Andrew Gomes*
Department of Diagnosis and Nonvascular radiology, National Interventional Radiology Partners, USA
- *Corresponding Author:
- Andrew Gomes
Department of Diagnosis and Nonvascular radiology
USA
Tel: +20223494183
E-mail: agomes@nationalirpartners.com
Received date: April 26, 2018; Accepted date: May 17, 2018; Published date: May 24, 2018
Citation: Andrew Gomes (2018) The Accuracy of a Volume Plethysmography System as Assessed by Contrast Angiography. J Prev Med Vol.3 No.2: 15.
Copyright: © 2018 Gomes A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Conventional contrast angiography (CA) is the standard diagnostic modality for peripheral artery disease of the lower extremities. However, CA presents possible risks, including contrast nephropathy and radiation exposure. An invasive procedure, it is not chosen to evaluate non-invasive physiologic procedures testing for vascular disease. Alternatively, Duplex ultrasonography (Duplex) is used to assess other non-invasive procedures that evaluate the presence or extent of peripheral artery disease. However, Duplex is not an infallible tool. Rather, another non-invasive physiologic test for peripheral artery disease is a volume plethysmography system (VPS), a blood volume waveform visualization and evaluation tool.
Objective: To determine the accuracy of a VPS as an aid for the primary care physician to more accurately identify patients with peripheral artery disease who might benefit from preventive management.
Design: Retrospective case series and a case presentation. Patients: Community vascular laboratory group consisting of 48 patients.
Methods and Findings: Patients were tested bilaterally with a VPS on all four limbs as per standard testing protocol. They subsequently underwent peripheral CA due to symptoms, physical examination, VPS or Duplex results suggestive of significant peripheral artery disease. Using CA as the gold standard for determining peripheral artery disease, the VPS had sensitivity of 86.0%, specificity of 100% and accuracy of 87.5%. Results indicated flow obstruction in 37 of 48 patients and showed no flow obstruction in 11 of 48 patients. The CA results confirmed peripheral artery disease in 37 of 37 patients with suspected peripheral artery disease by the VPS result.
Conclusion: Accuracy of the VPS is verified by CA results, supporting its use as an aid for practicing physicians to accurately diagnose peripheral artery disease. VPS is intended to provide primary care physicians with a tool, which in combination with clinical judgment, to more accurately identify patients with peripheral artery disease who might benefit from preventive management. When appropriate, patients may be referred to a vascular specialist.
Keywords
Peripheral artery disease; Hypertension; Cardiovascular risk factor; Contrast angiography; Duplex ultrasonography; plethysmography; Proximal popliteal artery
Abbreviations
CA: Contrast Angiography; VPS: Volume Plethysmography System
Introduction
Conventional contrast angiography (CA) is considered the standard diagnostic modality for peripheral artery disease of the lower extremities. Possible risks of CA include hematoma, contrast nephropathy and radiation exposure. Because it is invasive, CA is generally not chosen to evaluate non-invasive physiologic procedures that test for vascular disease. Instead, Duplex ultrasonography (Duplex) is generally used to assess the accuracy of other non-invasive techniques which are designed to evaluate the extent or presence of peripheral artery disease.
However, Duplex is not an infallible tool. RW Franz et al. [1] reviewed a total of 373 lesions in 278 patients who had undergone Duplex followed by CA. Using CA as the gold standard; Duplex demonstrated a sensitivity of 79.7% and a specificity of 79.2%. Duplex velocities and the angiographic stenosis severity correlated in 70.8% of lesions. In the 27 cases of highly stenotic disease documented by CA where Duplex was a false negative, the location of 18 or 66.7% were below the knee.
Another non-invasive physiologic test for peripheral artery disease is a blood volume waveform visualization and evaluation tool that performs volume plethysmography on multiple segments of the arterial anatomy. Known as volume plethysmography system (VPS), it is used in the primary care office to identify patients at risk for cardiovascular events and aids in the earlier diagnosis of peripheral artery disease. As an example, a case report is described herein, where this type of technology initiated a cascade of activity that led to preservation of cardiac function and treatment of peripheral artery disease.
VPS has been demonstrated [2] to be more accurate than ankle-brachial index measured by the Doppler-based blood pressure cuff (cuff Doppler). The basis for determining accuracy for VPS was to do three tests (VPS, cuff Doppler, and Duplex) on all subjects in the study population [2] and to employ Duplex as the gold standard. The purpose of this analysis was to assess the sensitivity, specificity and accuracy of VPS to determine the presence or absence of flow obstruction when using CA as the gold standard instead of Duplex.
Methods
At a primary care practice’s community vascular laboratory group, a total of 48 patients* were tested bilaterally with VPS on all four limbs as per standard testing protocol, and subsequently with peripheral CA. The reason for CA was due to symptoms, physical examination, or results of VPS or Duplex tests suggestive of significant peripheral artery disease (N=41 patients), or clinical suspicion of significant coronary artery disease (N=7 patients), which is often associated with peripheral artery disease [3-5]. Of the 48 patients, 37 also had Duplex performed prior to the CA.
The VPS measurements were performed bilaterally at the lower and upper extremities with the QuantaFlo™ (Semler Scientific, Inc., San Jose, CA). A sensor was placed on a single digit of each extremity in sequential fashion. The sensor included a transducer, which detected changes in arterial blood volume. During each 15-second measurement, the sensor detected reflected infrared light and recorded the blood pulse volume changes in the arterial segments (“pulse volume recording” or “volume plethysmography”).This signal is digitized and sent to a computer, which ran a specifically-designed software application. The application calculated the result via a proprietary algorithm, which is based on the features of the volume plethysmography signals from the segmental measurements.
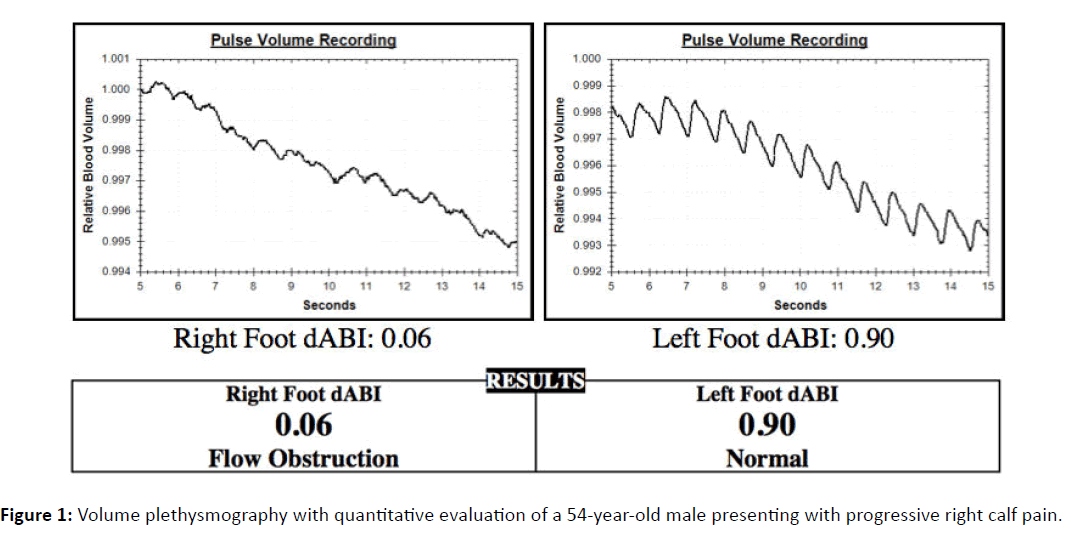
The system software determined the degree of flow obstruction in each lower extremity relative to the upper extremities. The calculated value was a ratio or index of leg to arm pulse volume analyses. Standard threshold values for flow obstruction were no abnormality of >0.99, mild >0.90, moderate >0.60, significant >0.30 and severe =<0.30. Data storage software captured the report form (Figure 1) and enabled analysis of all records of testing in the system archive.
All patient data were de-identified. VPS determined the presence or absence of flow obstruction. The interpretation of the Duplex or CA in terms of the presence of absence of peripheral artery disease was determined by the physician specialist attending to the patient as recorded on the procedure report form. In each case from the study, the VPS system was able to perform the measurement and analysis. There were no limbs that could not be measured.
The case description is an additional patient contributed by a vascular surgeon who uses VPS in his specialist practice and it illustrates the cardiovascular risk of myocardial infarction and stroke associated with the finding of peripheral vascular disease.
This article is a retrospective analysis of de-identified case studies that reside in a database archive. No private information was accessed. No identities were shared. As such, there was no requirement to obtain Institutional Review Board approval and no application for such approval was submitted.
Results
The clinical characteristics are shown in Table 1. A large proportion of this patient cohort had multiple cardiovascular risk factors, and many had histories of interventional procedures for peripheral artery disease. Approximately half had classic symptoms of claudication or rest pain, and one-fifth had lower extremity ulcers or non-healing sores. There was a slight preponderance of men over women. The average patient age was 68.5 years.
| Clinical data | (N=48) |
|---|---|
| Claudication | 52.10% |
| Rest pain | 50.00% |
| Non-healing wound | 20.80% |
| Hyperlipidemia | 66.70% |
| Hypertension | 81.30% |
| Diabetes | 70.80% |
| History of peripheral intervention | 37.50% |
| History of amputation | 14.60% |
| History of bypass graft of peripheral artery | 4.20% |
Table 1: Patients presenting clinical characteristics.
As demonstrated in Table 2, VPS results indicated flow obstruction in 37 of 48 patients and showed no flow obstruction in 11 of 48 patients. The CA results confirmed 37 of 37 patients who were suspected of peripheral artery disease by the VPS results. The CA results confirmed no peripheral artery disease in five of the 11 patients who had no flow obstruction on the VPS.
| VPS | |
| True positive results | 37 |
| False positive results | 0 |
| True negative results | 5 |
| False negative results | 6 |
| Total | 48 |
| Sensitivity | 86.00% |
| Specificity | 100.00% |
| Accuracy | 87.50% |
Table 2: Patients tested with VPS and CA (N=48).
Using CA as the gold standard for determining peripheral artery disease, VPS had a sensitivity of 86.0%, specificity of 100% and accuracy of 87.5%.
In this study, 37 of the 48 patients also had a Duplex performed. The VPS and Duplex results agreed in 28 of 37 patients. In the 28 cases of agreement, both non-invasive techniques identified PAD in 27 cases and no peripheral artery disease in one case. All 28 cases were confirmed by CA. In the nine cases of disagreement between the non-invasive techniques, the VPS reading demonstrated no significant flow obstruction and Duplex indicated that peripheral artery disease was present. The CA result agreed with VPS in three cases and agreed with Duplex in six cases.
Case Presentation
An active, 54-year-old male, without a significant past medical history, was referred for evaluation. Symptoms included a twoweek history of progressive right calf pain. Patient risk factors include controlled hypertension, hyperlipidemia, Type II diabetes, and a remote 30-year smoking history.
Volume plethysmography with quantitative evaluation was performed on all four limbs. As viewed in Figure 1, the left lower extremity was normal and the right lower extremity showed severe flow obstruction. Duplex ultrasound confirmed disease in the right proximal popliteal artery.
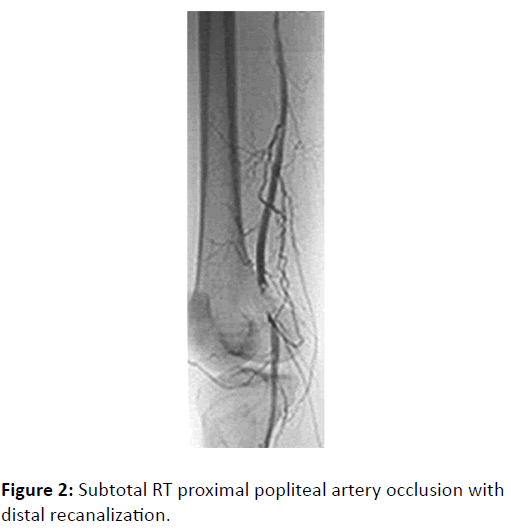
The pre-procedure EKG for a CA and embolectomy was abnormal and led to an urgent coronary arteriography. Findings were occlusions of the mid right coronary artery and proximal 1st diagonal artery, which were treated with angioplasty and stenting. CA of the lower extremities showed subtotal right proximal popliteal artery occlusion with distal recanalization, which was treated with embolectomy (Figure 2). The patient was stable throughout and no myocardial damage was noted during his hospitalization.
Discussion
In studies where Duplex scans judge the accuracy of the VPS results: it was expected [2] that 85-90% of VPS tests that are positive for flow obstruction are confirmed by Duplex. It is unclear that Duplex is always correct when it disagrees with VPS. CA is well established to be a superior diagnostic tool to Duplex [1,6] and the data presented here showed that CA confirmed that all 37 patients with flow obstruction on their VPS test actually had peripheral artery disease. There were no false positive VPS results using CA as the gold standard. It is conceivable that when there is disagreement between a positive VPS result and a normal Duplex test, it might be that the Duplex scan was a false negative and not that the VPS was a false positive.
Of the 11 patients that had a VPS result of no flow obstruction, the CA findings confirmed in five cases that VPS results were correct. Using CA as the gold standard, VPS demonstrated a sensitivity of 86.0%, specificity of 100%, and accuracy of 87.5%.
This study is limited by a small sample size and there is no intent to make any conclusions about the comparative clinical value of VPS and Duplex. The two methodologies are complementary techniques. VPS is relatively inexpensive, fast, and easy-to-use, so it is usually deployed in the primary care office and operated by a medical aide. The primary care provider refers cases of significant flow obstruction detected by VPS to a vascular specialist for further testing. Duplex requires a vascular lab and a vascular technologist to perform, so it is usually the next non-invasive test to evaluate patients suspected to have peripheral artery disease. Duplex provides anatomical location of disease in many cases and is utilized prior to the invasive CA in cases where intervention is considered likely and beneficial.
It is a general recommendation to use the VPS as an aid in the diagnosis of peripheral artery disease, especially when physical examination and history are unremarkable, as many patients with peripheral artery disease are asymptomatic and/or have no abnormal findings on physical examination [7].
The case presentation illustrates the systemic nature of atherosclerosis and the importance of identifying peripheral artery disease as a risk factor for coronary artery disease. Previous studies have shown that patients with peripheral artery disease suffer a 21% chance of myocardial infarction, cerebrovascular accident, hospitalization, or mortality within one year of diagnosis [3]. Early preventive measures might improve prognosis for these patients.
Conclusion
The accuracy of the VPS test is verified herein by CA results, supporting the use of VPS as an aid to the practicing physician to accurately diagnose peripheral artery disease. VPS is intended to provide the primary care physician with a tool that in combination with clinical judgment will more accurately identify patients with peripheral artery disease who might benefit from preventive management. When appropriate, patients may be referred to a vascular specialist.
Acknowledgement
John B Long, MD Department of Vascular Surgery, BASS Medical Group, Daniel Burnham Court. Dr. Long has agreed to contribute the above case presentation to this original research paper. The authors’ views expressed are his or her own and not an official position of the institution or funder.
Conflicts of Interest
Dr. Gomes has no conflict of interest. Dr. Long has no conflict of interest.
References
- Franz RW, Jump MA, Spalding MC, Jenkins JJ (2013) Accuracy of duplex ultrasonography in estimation of severity of peripheral vascular disease. Int J Angiol 22: 155.
- Schaefer ME, Long JB, Pollick C (2016) Non-invasive detection of vascular disease in the arteries of the lower extremity: Clinical evaluation of PVS compared to doppler and definitive imaging.
Vasc Dis Management March 2016 supplement - Steg PG, Bhatt DL, Wilson PW, D’Agostino R, Ohman EM, et al. (2007) One year cardiovascular event rates in outpatients with atherothrombosis. JAMA 297: 1197-1206.
- Hertzer NR, Beven EG, Young JR, O'hara PJ, Ruschhaupt WF, et al. (1984) Coronary artery disease in peripheral vascular patients: A classification of 1000 coronary angiograms and results of surgical management. Ann Surg 199: 223.
- Kownator S, Cambou JP, Cacoub P, Léger P, Luizy F, et al. (2009) Prevalence of unknown peripheral arterial disease in patients with coronary artery disease: Data in primary care from the IPSILON study. Arch Cardiovasc Dis 102: 625-631.
- Polak JF, Karmel MI, Mannick JA, O'Leary DH, Donaldson MC, et al. (1990) Determination of the extent of lower-extremity peripheral arterial disease with color assisted duplex sonography: Comparison with angiography. AJR Am J Roentgenol 155: 1085-1089.
- Greenland P, Alpert JS, Beller GA (2010) ACCF/AHA guideline for assessment of cardiovascular risk in asymptomatic adults: A report of the american college of cardiology foundation/american heart association task force on practice guidelines. J Am Coll Cardiol 56: 50-103.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences